Structure and Function
Figure 3: a1/a2 domain of HLA-B2705. Peptide binding cleft is blue, bound nonapeptide is yellow.
Figure 3: a1/a2 domain of HLA-B2705. Peptide binding cleft is blue, bound nonapeptide is yellow.
Domain Structure
MHC I molecules consist of two chains: a and b. The a chain has 3 90-amino-acid domains, a1-a3, encoded by region A, B and C genes in the HLA complex (see Table 1). The a3 domain contains a 40-amino-acid hydrophobic transmembrane segment (which anchors the molecule into the cell membrane) and a 30-amino-acid cytoplasmic tail (Voet and Voet, 1995). Each a domain contains roughly 90 amino acids. Not shown are the a3 and b2-microglobulin domains, which catalyze the initial assembly of the MHC molecule and are organized into an immunoglobulin fold (2 b sheets formed by antiparallel b strands) highly conserved among class I MHC molecules.
The peptide binding cleft is composed of the a1 and a2 domains, which interact to form a base of 8 b sheets with a helices running along either side (Figure 3). The cleft is approximately 25 angstroms long, 10 angstroms high and 11 angstroms wide, accomodating peptides of 8-10 amino acids (Kuby, 1997).
Conserved Residues and Binding
Nine conserved residues exist at each end of the peptide binding cleft: Tyr-7, Tyr-59, Asp-77, Tyr-84, Thr-143, Lys-146, Trp-147, Tyr-159, and Tyr-171 (Madden et al, 1992). These residues bind to conserved anchor residues in the nonapeptide: Arg-2, Ile-3, and Lys-9 (Figure 4).
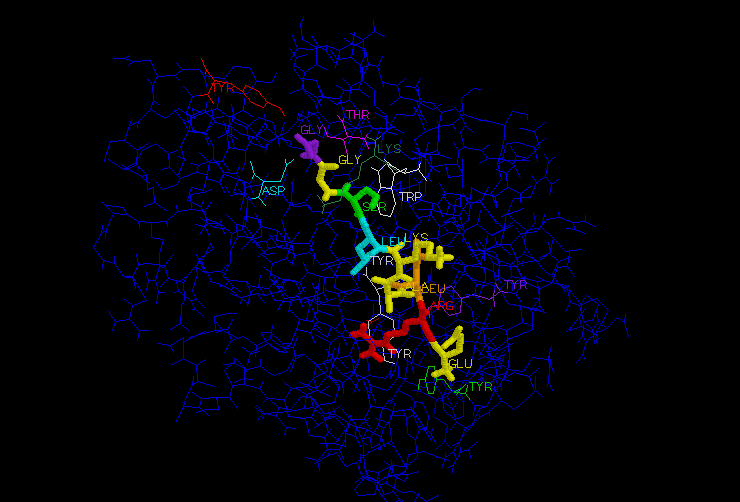
Figure 4: Conserved residues involved in peptide-MHC binding. MHC residues are shown in wire-frame, and labeled. The peptide (thicker molecule in center) has conserved residues which hydrogen bond with conserved MHC residues, anchoring the peptide into the cleft and stabilizing the MHC.The carboxy terminus of peptide residue 1 (not conserved) forms a hydrogen bond with MHC residue Tyr-159 (conserved, shown in green at bottom of figure 4). The amino group of peptide residue 1 also forms a hydrogen bond network with MHC residues Tyr-7, Tyr-59, and Tyr-171 (all conserved. Shown in white, white, and purple in figure 4) (Madden et al, 1992). ***See also figure 5 for alternate representation of residues involved in binding.
Peptide residue 8 (not conserved, serine in figure 4) forms a hydrogen bond with conserved MHC residue Trp-147 (shown in white). This interaction forces peptide residue 9 (glycine, conserved, shown in purple) down into the cleft where it serves as an anchor residue.
A unique feature of the HLA-B2705 peptide cleft is an arginine-binding pocket. Peptide residue Arg-2 (shown in red in figure 4) is highly conserved, and fits into a pocket formed by 4 non-conserved MHC residues, forming a planar network of hydrogen bonds. This arginine-binding pocket is found in all of the known HLA-B27 molecules, but not in the other HLA molecules (Madden et al, 1992). Some of the latter have a similar pocket in the same location, but with residues different enough to significantly change the character of the pocket, such as substitution of phenylalanine for cysteine.
Figure 5 MHC-peptide binding. Conserved residues are colored in the same scheme used in Figure 4 (peptide begins with cyan Glu 1, ending with purple Gly 9). Peptide residues not involved in hydrogen bonding are colored yellow. The left half of figure 5 is oriented to show the residues involved in hydrogen binding at the N-terminus (peptide runs N to C from left to right), and the right half is oriented to show those involved at the C terminus (peptide runs C to N from bottom to top).
.
The binding energy present at the ends of the peptide-binding cleft appears to be essential to the ability of an MHC I molecule to bind a large number of different peptides. Peptide residues 1-3, 8 and 9 provide most of the peptide-MHC interaction (figures 5 and 6), and these residues make 3/4 of all possible hydrogen bonds, using their binding potential almost completely. Anchor side chains in the MHC then interact with non-conserved residues in the middle of the peptide chain, positioning the peptide correctly in the cleft.

Figure 6 Ribbon diagram of a1 -- a2 domain of MHC I with bound nonapeptide. Color scheme is the same used in figures 4 and 5, conserved MHC residues involved in hydrogen bonding are colored and labeled, peptide residues involved in binding are also colored (all peptide residues are labeled). This view provides a clearer picture of the hydrogen-bonding residues in the context of the rest of the peptide-binding domain. Also visible is the arching of the middle of the peptide away from the cleft's floor, and the lack of MHC residues available for binding in this region.
Peptide binding by MHC I is necessary for stable expression of the MHC molecule on the cell surface. Peptides bind MHC I with high affinity: the dissociation constant KD is approximately 10-6 (Kuby, 1997). The association rate is not very fast, but the dissociation rate is also very slow under physiologic conditions (compare to values in Table 2), indicating that most MHCs expressed on a cell at any given time will be associated with a peptide. Note that this value refers to the association between an MHC I molecule and a nonapeptide. Peptides as long as 13 amino acids have been isolated with MHC I molecules, but the half-life of the association is considerably shorter (Madden et al, 1992).
Interaction Kd Antibody-antigen interaction, high-affinity antbody 10-8-10-10 DNA-binding protein with specific site 10-8-10-10 Antibody-antigen interaction, weak antibody 10-6 Enzyme-substrate interaction 10-4-10-10 Table 2. Dissociation constants for biologically significant interactions (from Brent, 1997).
Although most known peptides bound by MHC I molecules are nonameric, the configuration in which the peptide binds allows for variation. While the ends of the peptide are anchored in the ends of the binding cleft, the middle of the peptide arches away from the b sheet floor, which may facilitate presentation to TCRs on cytotoxic T cells. A longer peptide could therefore be accomodated by arching more, and a shorter peptide could likewise be more extended and still form all of the anchor residue interactions (Kuby, 1997). This also increases the variability of peptides that an MHC molecule can bind, because the peptide residues in the middle of the chain generally do not contact the MHC binding cleft, and are not necessary for specific associations.